- 翰林提供學術活動、國際課程、科研項目一站式留學背景提升服務!
- 400 888 0080
2009 AP Chemistry化學真題系列之簡答題Form B免費下載
歷年AP Chemistry化學系列
真題與答案下載

翰林國際教育全網首發
力爭超快速發布最全資料
助你在升學路上一帆風順
為你的未來保駕護航
2009 AP Chemistry Free-Response Questions (Form B)Free Download
2009 AP 化學簡答題 Form B部分免費下載
考試時會提供花常用的等式與常量
以及化學元素周期表
此套Section II試卷共6題
每道大題含有不同數量的小題
共計時95分鐘
Part A計55分鐘,第1-3題各20分
Part B計40分鐘,第四題值10分,第5第6題各15分
部分真題預覽:
Section II, Part A,YOU MAY USE YOUR CALCULATOR FOR PART A可使用計算器:
![]()
2)A student performed an experiment to investigate the decomposition of sodium thiosulfate, Na2S2O3 , in acidic solution, as represented by the equation above. In each trial the student mixed a different concentration of sodium thiosulfate with hydrochloric acid at constant temperature and determined the rate of disappearance of S2O32?(aq). Data from five trials are given below in the table on the left and are plotted in the graph on the right.
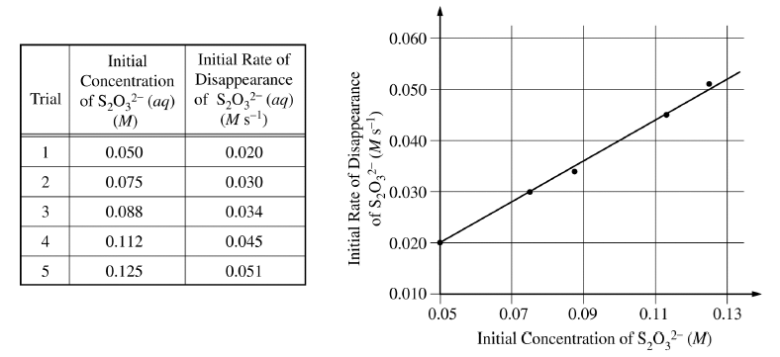
- Identify the independent variable in the experiment.
- Determine the order of the reaction with respect to S2O32-. Justify your answer by using the information above.
- Determine the value of the rate constant, k, for the reaction. Include units in your answer. Show how you arrived at your answer.
- In another trial the student mixed 0.10 M Na2S2O3 with hydrochloric acid. Calculate the amount of time it would take for the concentration of S2O32- to drop to 0.020 M.
- On the graph above, sketch the line that shows the results that would be expected if the student repeated the five trials at a temperature lower than that during the first set of trials.
2009 AP Chemistry化學 FRQ微積分(Form B)完整版答案免費下載
請持續關注,稍后更新
2009 AP Chemistry Free-Response Questions Free Download
2009 AP 化學簡答題部分免費下載
考試時會提供花常用的等式與常量
以及化學元素周期表
此套Section II試卷共6題
每道大題含有不同數量的小題
共計時95分鐘
Part A計55分鐘,第1-3題各20分
Part B計40分鐘,第四題值10分,第5第6題各15分
下載方式請點擊下方鏈接
完整版真題資料可以底部二維碼免費領取↓↓↓
[vc_btn title="查看更多AP Chemistry化學課程相關詳情" color="primary" align="center" i_icon_fontawesome="fa fa-globe" css_animation="zoomIn" button_block="true" add_icon="true" link="url:http%3A%2F%2Fwww.linstitute.net%2Farchives%2F25860||target:%20_blank|"]

最新發布
? 2025. All Rights Reserved. 滬ICP備2023009024號-1









